The Osmosis and Tonicity Worksheet is a great tool to help students understand the concepts of osmosis and tonicity. Osmosis is the movement of water across a semi-permeable membrane, while tonicity is the measure of the concentration of solutes in a solution relative to a reference solution. This worksheet will help students learn how osmosis and tonicity work, and how to apply them to everyday life. It will also help them to understand the effects of different concentrations of solutes on the movement of water. With this worksheet, students will be able to analyze and interpret data related to osmosis and tonicity.
Exploring the Different Types of Osmosis and Tonicity Worksheets
Osmosis and tonicity are two important concepts in biology that are necessary to understand when studying cell physiology. Osmosis is the diffusion of water across a membrane while tonicity is the ability of a solution to cause cells to shrink or swell. Worksheets are an effective way to help students learn and retain information about osmosis and tonicity. Different types of osmosis and tonicity worksheets can be used to help students better understand these two concepts.
The first type of worksheet is a basic knowledge worksheet. This worksheet helps students learn the basics of osmosis and tonicity. It includes simple questions about the process of osmosis and tonicity and how they both work. This type of worksheet is great for students who are just learning about these concepts and need to gain an understanding of their basic workings.
Contents
- 0.1 Exploring the Different Types of Osmosis and Tonicity Worksheets
- 0.2 Understanding Osmosis and Tonicity Through Real-World Examples
- 0.3 Creating an Effective Osmosis and Tonicity Worksheet for Your Classroom
- 0.4 Understanding How Osmosis and Tonicity Affect Cells and Organisms
- 0.5 Analyzing Osmosis and Tonicity Worksheets and the Processes They Represent
- 0.6 Images of Osmosis And Tonicity Worksheet With Answer Key
- 0.7 Download Osmosis And Tonicity Worksheet With Answer Key
- 1 Conclusion
The second type of worksheet is an application worksheet. This worksheet requires students to apply the concepts of osmosis and tonicity to real-world problems. This type of worksheet is great for more advanced students who have already gained a basic understanding of osmosis and tonicity. It can also help students to understand how these concepts can be used to solve real-world problems.
The third type of worksheet is a comparison worksheet. This worksheet helps students compare the different types of osmosis and tonicity and how they differ from one another. This type of worksheet can help students to better understand the differences between the two concepts and how they can be used in different scenarios.
Finally, the fourth type of worksheet is a practice worksheet. This worksheet helps students practice using the concepts of osmosis and tonicity. This type of worksheet is great for students who have already gained a basic understanding of the two concepts and need to practice using them in different scenarios.
Overall, there are many different types of osmosis and tonicity worksheets available for students to use. These worksheets can help students learn and retain information about these two important concepts. They can also help students apply the concepts to real-world problems or compare the different types of osmosis and tonicity.
Understanding Osmosis and Tonicity Through Real-World Examples
Osmosis is a process by which liquids, gases, and dissolved solids are transferred from an area of high concentration to an area of low concentration. Tonicity is a measure of the ability of a solution to cause a cell to gain or lose water. Both concepts are important in understanding the mechanisms of cellular respiration and transport.
To better understand osmosis and tonicity, consider a real-world example. Imagine a beaker filled with a solution of salt water. The salt molecules will, due to their size and charge, dissolve in the water and become evenly distributed throughout the solution. This creates a solution of equal concentration on both sides of the beaker, meaning the concentration of salt is the same in both the water and the beaker.
Now, imagine a semi-permeable membrane is placed in the beaker. This membrane will allow water to pass through, but not the salt molecules. This will create two distinct solutions on either side of the membrane, one with a higher concentration of salt and one with a lower concentration.
The higher concentration of salt in one side of the beaker will act as an osmotic pressure, drawing water molecules from the lower concentration side. This is osmosis in action, as the water is being transferred from an area of low concentration to one of high concentration.
The tonicity of the solution can be determined by measuring the amount of water that is transferred across the membrane. If the amount of water transferred is equal to the amount of salt on the high-concentration side, then the solution is said to be isotonic. If the amount of water transferred is less than the amount of salt on the high-concentration side, then the solution is said to be hypertonic. Conversely, if the amount of water transferred is greater than the amount of salt on the high-concentration side, then the solution is said to be hypotonic.
Understanding osmosis and tonicity is essential for understanding the mechanisms of cellular respiration and transport. Through examining a real-world example, we can gain a better understanding of how these two concepts work together to facilitate the movement of molecules within a solution.
Creating an Effective Osmosis and Tonicity Worksheet for Your Classroom
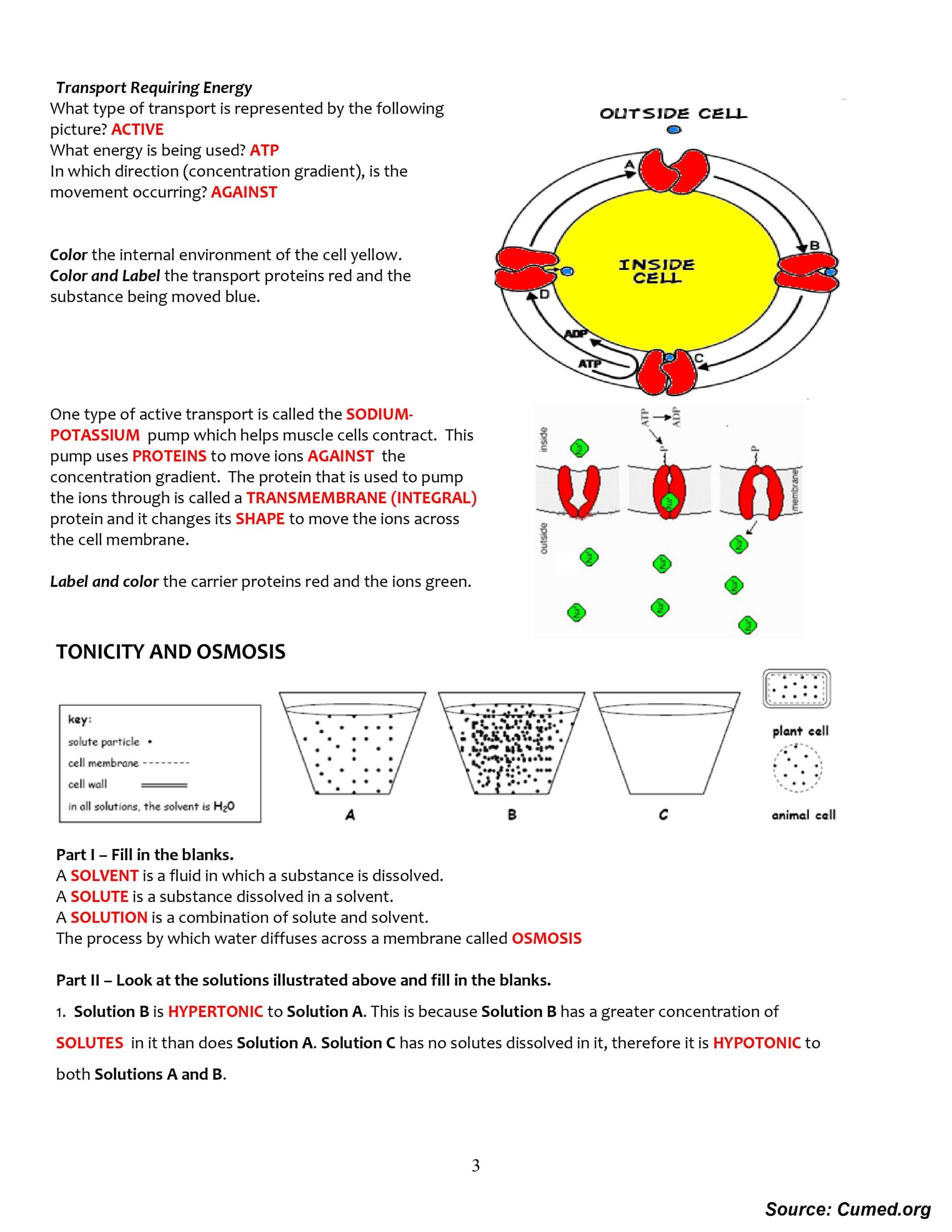
Osmosis and Tonicity Worksheet
Instructions: Answer the following questions to the best of your ability.
1. What is osmosis?
Osmosis is the diffusion of water molecules from an area of higher water concentration to an area of lower water concentration through a semi-permeable membrane.
2. What is tonicity?
Tonicity is the ability of a solution to cause a cell to gain or lose water depending on the relative concentrations of solutes inside and outside the cell.
3. What is hypertonic?
Hypertonic is a solution that has a higher concentration of solutes outside the cell than inside the cell. This causes the cell to lose water, resulting in it shrinking.
4. What is hypotonic?
Hypotonic is a solution that has a lower concentration of solutes outside the cell than inside the cell. This causes the cell to gain water, resulting in it swelling.
5. What is isotonic?
Isotonic is a solution that has the same concentration of solutes inside and outside the cell. This causes the cell to remain the same size.
6. What are the three factors that affect osmosis and tonicity?
The three factors that affect osmosis and tonicity are the concentration of solutes, the permeability of the cell membrane, and the size of the solutes.
7. Describe the process of osmosis.
The process of osmosis is the diffusion of water molecules from an area of higher water concentration to an area of lower water concentration through a semi-permeable membrane. Water molecules move from an area of higher concentration to an area of lower concentration in order to balance the concentration of solutes on both sides of the membrane. This process continues until the solute concentrations on both sides of the membrane are equal.
Understanding How Osmosis and Tonicity Affect Cells and Organisms
Osmosis and tonicity are important concepts when considering the behavior of cells and organisms. Osmosis is the diffusion of water across a selective permeable membrane, such as the cell membrane, from an area of low solute concentration to an area of higher solute concentration. The movement of water in osmosis is driven by the concentration gradient between two solutions. Tonicity refers to the ability of a solution to cause a cell to shrink or swell, depending on its relative concentration of solutes compared to the cell.
The concentration gradient that drives osmosis is critical to the survival of cells and organisms. If a cell is placed in a solution with a higher solute concentration than the cell, the water will move out of the cell and the cell will shrink. This is known as “hypertonic”. Conversely, if a cell is placed in a solution with a lower solute concentration than the cell, water will move into the cell, causing it to swell. This is known as “hypotonic”.
In an organism, osmosis and tonicity have important implications for the health and functioning of cells. For example, in plants, osmosis is necessary for water to move from the roots to the leaves, allowing for photosynthesis to occur. In animals, osmosis is necessary for the transport of nutrients, electrolytes, and other molecules from the blood and other bodily fluids to the cells. In both plants and animals, the tonicity of the environment is important for cells to maintain their normal volume and shape, and thus to be able to properly perform their metabolic and structural functions.
Osmosis and tonicity are also essential to the survival of aquatic organisms, such as fish. Aquatic organisms live in an environment with a constantly changing tonicity, and must be able to adapt to these changes in order to survive. Osmosis plays a crucial role in this process, allowing them to regulate the concentration of solutes inside and outside of their cells by absorbing or excreting water.
In conclusion, osmosis and tonicity are fundamental concepts in the study of cells and organisms. Osmosis is the diffusion of water across a selective membrane, driven by the concentration gradient between two solutions, while tonicity is the ability of a solution to cause a cell to shrink or swell. Osmosis and tonicity play important roles in the metabolism and functioning of cells, and in the survival of organisms in aquatic environments. Understanding these concepts is critical to gaining a better understanding of the behavior of cells and organisms.
Analyzing Osmosis and Tonicity Worksheets and the Processes They Represent
Osmosis and tonicity are important concepts to understand in the biological sciences, as they explain how water moves across cell membranes. Worksheets which analyze these processes are used in classrooms to help students gain a better understanding of how these concepts work.
Osmosis is a process by which water molecules move from an area of higher water potential to an area of lower water potential, through a partially permeable membrane. Tonicity is the measure of how much solute is dissolved in a given solution, relative to another solution. The tonicity of a solution can be compared to another solution, usually a cell membrane.
Osmosis and tonicity worksheets typically contain diagrams of cells and solutions, along with questions that require the student to analyze the diagrams and determine the direction of water flow. In some worksheets, the student is also asked to calculate the osmotic pressure of the solutions. These worksheets help students understand the concept of osmosis and tonicity as it applies to living cells and their environment.
The worksheets also help students to develop their critical thinking skills, as they have to draw logical conclusions from the diagrams and the questions posed. Additionally, the questions require students to compare and contrast different solutions and their properties, which helps the student to better understand the concepts.
Osmosis and tonicity worksheets are a valuable tool in helping students to understand the concepts of osmosis and tonicity. By using these worksheets, students are able to gain a better understanding of how water moves across cell membranes and how the tonicity of a solution affects its environment. The worksheets also help to develop critical thinking and analytical skills, as students must draw logical conclusions from the diagrams and questions posed.
Images of Osmosis And Tonicity Worksheet With Answer Key




Download Osmosis And Tonicity Worksheet With Answer Key
Download Osmosis And Tonicity Worksheet With Answer Key: click here
Conclusion
This worksheet has provided a great overview of osmosis and tonicity, and how they are related. By understanding the concepts and the different types of solutions, students can now better understand how and why certain substances move in and out of cells. With this knowledge, students can apply these principles to real-life situations and make informed decisions about their health and nutrition.
